Food and Drug Administration approves Opdivo® (nivolumab) as adjuvant treatment of completely resected esophageal or gastroesophageal junction cancer in patients who have received neoadjuvant chemoradiotherapy. In the CheckMate-577 trial, we saw a doubling in median disease-free survival compared to placebo, which suggests that Opdivo could become a new standard of care for these patients. This is exciting news, providing renewed hope,” Kelly added.ġ. “Even after neoadjuvant CRT followed by surgery, there may be a high risk of recurrence for patients who do not achieve a pathologic complete response. 
These events were also observed with the placebo in 1% of patients each. The most common grade 3/4 AEs observed with nivolumab were pneumonitis and rash, which occurred in 1% of patients each. The most common AEs observed in the nivolumab arm included fatigue, diarrhea, pruritus, and rash, and diarrhea and fatigue were most common in the placebo arm. Treatment discontinuations were seen in 9% of the experimental arm compared with 3% of the placebo arm. These TRAEs were grade 3/4 in 13% of the nivolumab arm compared with 6% of the placebo arm. Treatment-related AEs were found to be more common in the nivolumab group compared with the placebo group. 
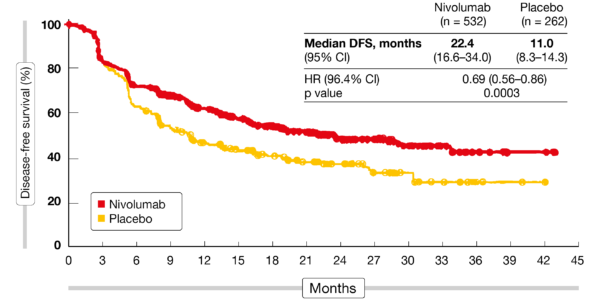
Grade 3/4 adverse events were observed in 34% of the adjuvant nivolumab arm compared with 32% of the placebo arm. Notably, 86% of the nivolumab-treated population had a 90% dose intensity. The safety analysis showed that adjuvant nivolumab and the placebo led to a similar amount of dose delays. In the subgroup of patients with squamous cell carcinoma who made up 29% of the overall study population, treatment with adjuvant nivolumab led to a median DFS of 29.7 months (95% CI, 14.4 to not evaluable) versus 11.0 months (95% CI, 7.6-17.8) in the placebo group (unstratified HR, 0.61 95% CI, 0.42-0.88). 001).įurther, an exploratory analysis of patients with adenocarcinoma who made up 70.9% of the overall study population, the median DFS was 19.4 months (95% CI, 15.9-29.4) among patients treated with nivolumab compared with 11.1 months (95% CI, 8.3-16.8) among those who receive placebo (unstratified HR, 0.75 95% CI, 0.59-0.96). 1,3Īt a median follow-up of 24.4 months, the median DFS observed with adjuvant nivolumab was 22.4 months (95% CI,16.6-34.0) compared with 11.0 months (95% CI, 8.3-14.3) in the placebo arm, achieved a 31% reduction in the risk of disease recurrence or death (HR, 0.69 95% CI, 0.56-0.85 P <. In addition to DFS, patients were evaluated for the secondary end points of overall survival and overall response rate. Placebo was administered at a matching dose level. Nivolumab was initially dosed at 240 mg every 2 weeks for 16 weeks, then was increased to 480 mg every 4 weeks. In the study, patients with resected esophageal or GEJ were randomized 2:1 to received either nivolumab (n = 532) or placebo (n = 262). Endowed Chair of Immunology at Baylor University Medical Center, in a statement. Kelly, MD, MBA, director, Baylor Scott & White Charles A. “Locally advanced esophageal and gastroesophageal junction cancers are aggressive tumor types that often require multiple approaches to address the disease, including chemotherapy, radiation and surgery,” said Ronan J. Following positive results in patients with melanoma, the finding from CheckMate-577 made esophageal or GEJ tumor the second tumor type to derive benefit from adjuvant nivolumab. The agent was approved based on findings from the phase 3, randomized, placebo-controlled, double-blind, multi-center CheckMate-577 trial, which demonstrated a statistically significant disease-free survival benefit with nivolumab when compared with placebo. The FDA granted approval to adjuvant nivolumab (Opdivo) for the treatment of completely resected esophageal or gastroesophageal junction (GEJ) cancer with residual pathologic disease in patients who have received neoadjuvant chemoradiotherapy (CRT), according to a press release from Bristol Myers Squibb, developer of the agent.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
